 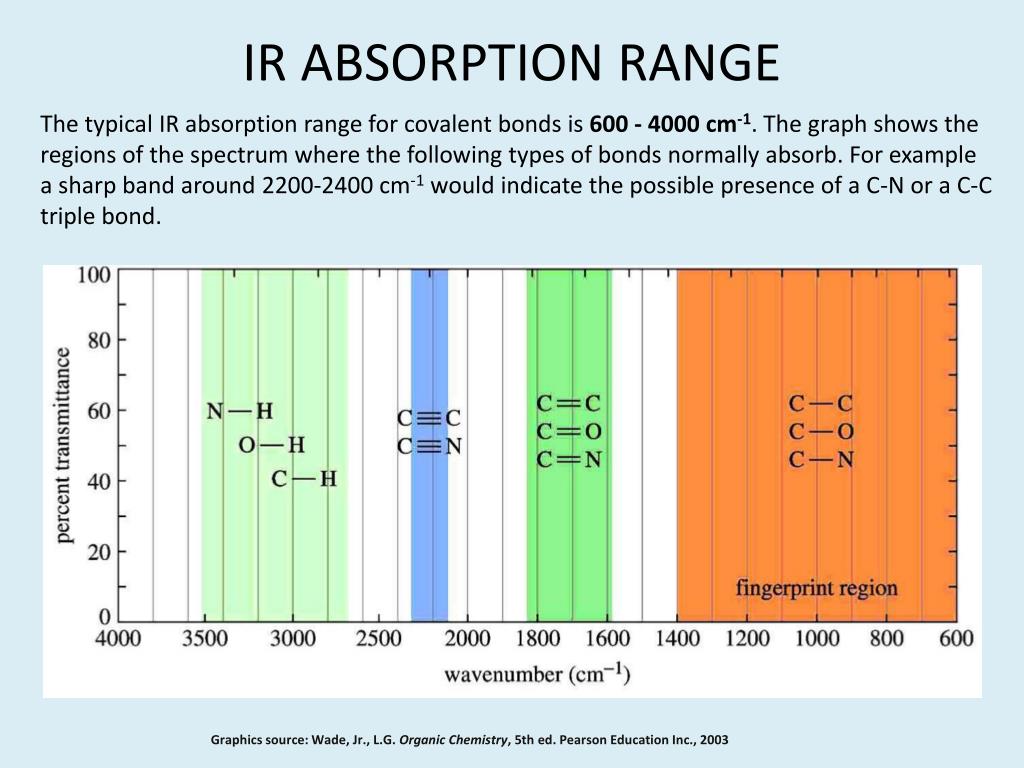
In addition to applying the SAC-IR spectroscopy to various applications, we are working to improve the sensitivity and expand the frequency range and increase the acquisition speed to meet the critical needs of protein characterization. By the patented solvent absorption compensation (SAC) technique, we can detect the amide I band of a protein solution with 100 times better sensitivity compared to conventional mid-IR spectroscopy. Recently, we have developed a new optical technique for quantum cascade laser (QCL)-based mid-IR absorption spectroscopy. Non-invasive optical characterization of low-concentration protein samples is critical for new types of drugs in their original formulation conditions. However, the interference by strong water absorption keeps FT-IR from characterizing low concentration samples of <10 mg/mL and prevents the usage of a long-pathlength optical cell. Extensive studies on FT-IR spectroscopy of proteins have shown that the amide bands can be used to semi-quantitatively characterize the secondary structure (e.g., a -helix and b -sheet) of the protein backbone conformation. FT-IR is advantageous because it does not require additional sample preparation steps, such as buffer exchange and dilution. Fourier-transform IR (FT-IR) technology is commonly used to characterize proteins and other biological molecules produced during biopharmaceutical processes. To ensure that no center of mass motion occurs, the center atom (yellow ball) will also move.Infrared (IR) absorption spectroscopy has been used widely as a non-invasive, label-free characterization method for complex biomolecules’ chemical identification and structure. Symmetric Stretch Asymmetric Stretch Twistingįigure 3: Types of Vibrational Modes. A CH 2 group is used as an example to illustrate stretching and bending vibrations below. Four bending vibrations exist namely, wagging, twisting, rocking and scissoring. A change in the angle occurring between two bonds is known as a bending vibration. High energy waves of light have short wavelengths and high frequencies. The electromagnetic spectrum in shown in Figure 5.4.1. In other words, it is the study of what happens to molecules in the presence of difference wavelengths of light. When there is a continuous change in the interatomic distance along the axis of the bond between two atoms, this process is known as a stretching vibration. Spectroscopy is the study of the interaction of matter with energy from the electromagnetic spectrum. This is not the case for large molecules due to several vibrational motions and interactions that will be experienced. These movements are easily defined for diatomic or triatomic molecules. Vibrational motions are defined by stretching and bending modes. 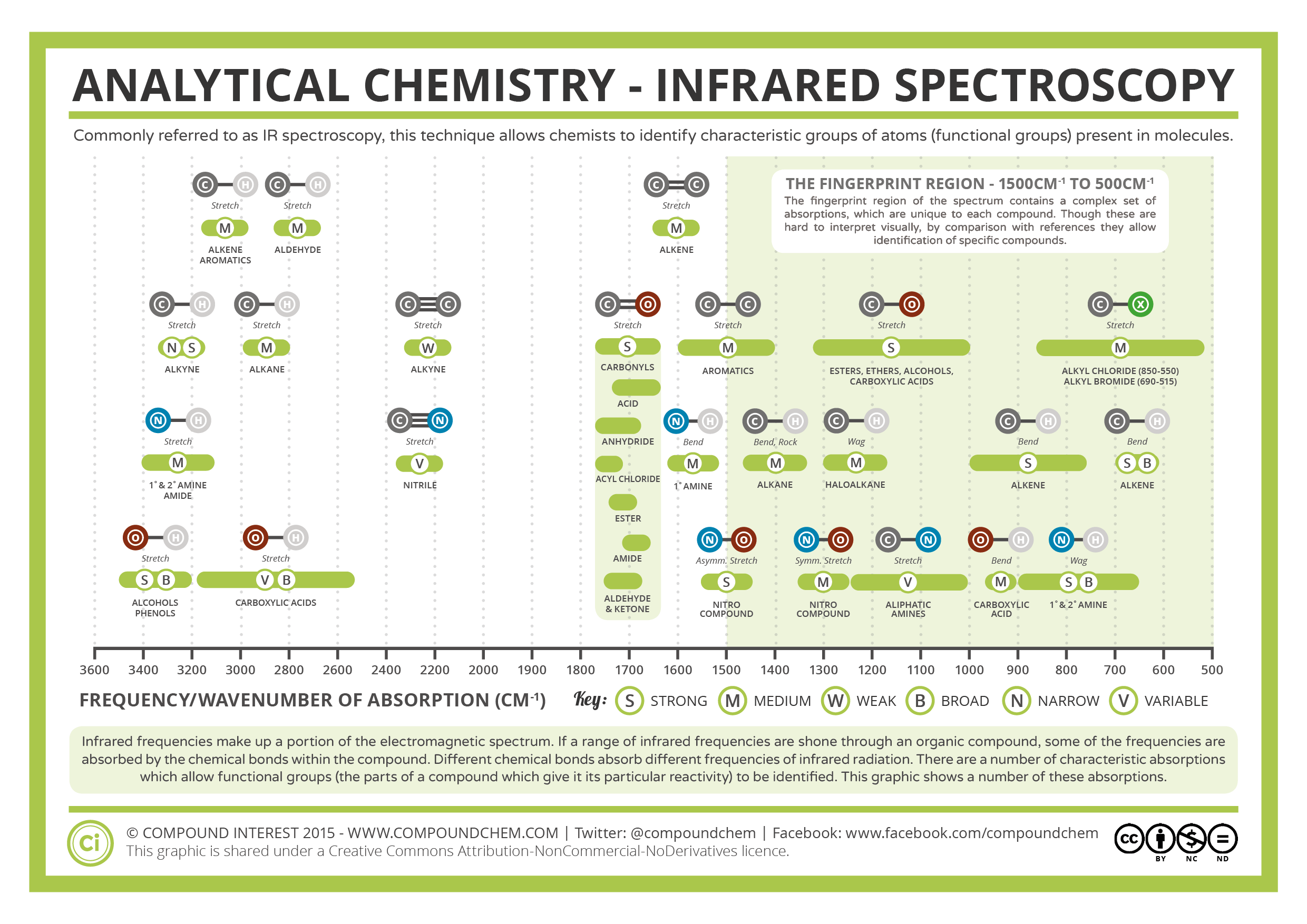
intensity of the transmission (or absorption). 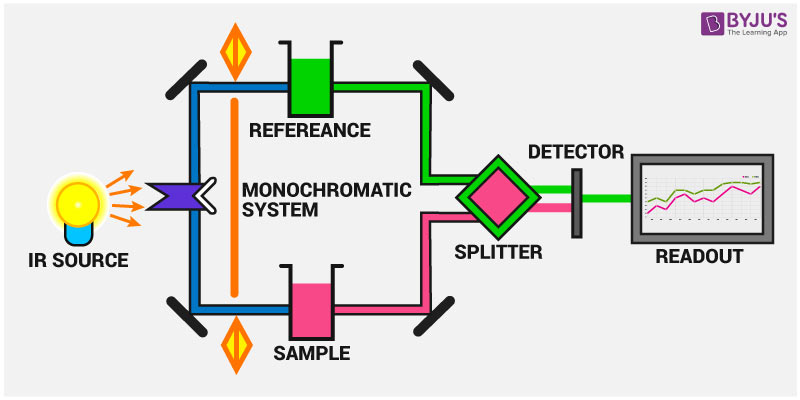
This causes the atom not to be stationary and to fluctuate continuously. The IR spectrum is basically a plot of transmitted (or absorbed) frequencies vs. The bond of a molecule experiences various types of vibrations and rotations. The fifth term represents the interaction between the vibration and rotational interaction of the molecule. These bands in IR spectra presented as wavenumber ( ). The second term represents anharmonicity and the fourth term represents centrifugal stretching. This absorption is quantized but vibrational spectra appear as bands rather than lines. The first and third terms represent the harmonicity and rigid rotor behavior of a diatomic molecule such as HCl. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
